- Registration Service of Medical Devices
- CRC services Medical Device Business License Medical Device Registration Contract Research Organization ISO 13485 Quality System Certification GMP CE Certification Service FDA Registration (Filing) Software Integrity Verification Service Production process validation service ISO 15378 Medical Package System Certification ISO 13485 Internal Auditors Training Regulatory Services For Medical Devices
- Information security service of medical
- ISO 27001 Information Security Certification ISO 20000 Information Service Certification Information system grade protection filing Business Continuity Management Services Internal Auditor Training
- Medical Software Development
- Software of Good Supply Practice(GSP) Production System Development Customized software development
- CONTACT US
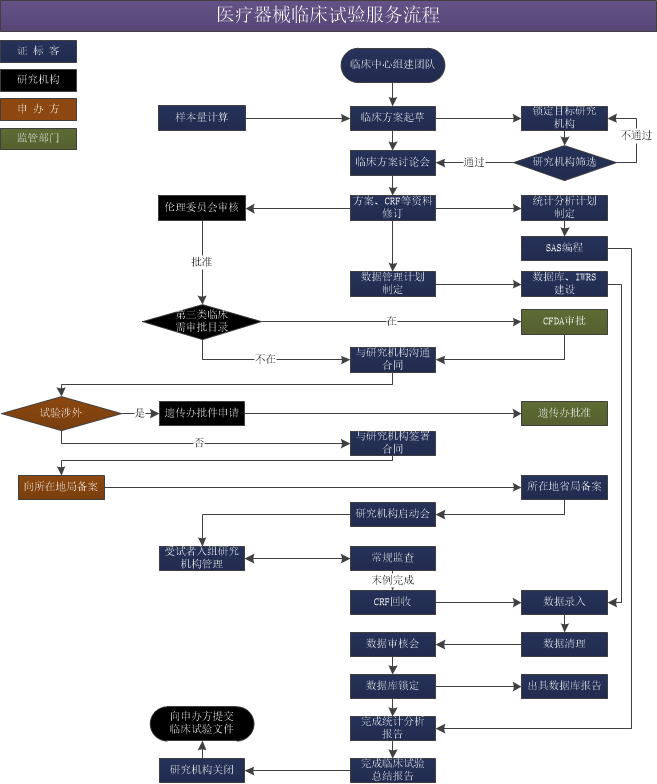
醫(yī)療器械臨床服務(wù)流程
CREATE TIME:2018-06-29 11:32BROWSE TIMES:2059
根據(jù)《醫(yī)療器械監(jiān)督管理?xiàng)l例》(國(guó)務(wù)院令第650號(hào))的規(guī)定,第Ⅰ類醫(yī)療器械實(shí)行產(chǎn)品備案管理,不需要進(jìn)行臨床試驗(yàn),但需要提交臨床評(píng)價(jià)資料;第Ⅱ、Ⅲ類醫(yī)療器械注冊(cè)時(shí),應(yīng)當(dāng)進(jìn)行臨床試驗(yàn)(免于進(jìn)行臨床試驗(yàn)的第Ⅱ類醫(yī)療器械目錄;免于進(jìn)行臨床試驗(yàn)的第Ⅲ類醫(yī)療器械目錄的醫(yī)療器械除外);免于進(jìn)行臨床試驗(yàn)的醫(yī)療器械,在進(jìn)行注冊(cè)申報(bào)時(shí),仍需提供臨床評(píng)價(jià)資料。
PREVIOUS: IVD clinical trial workflow chart
NEXT: NOTHING
NEXT: NOTHING